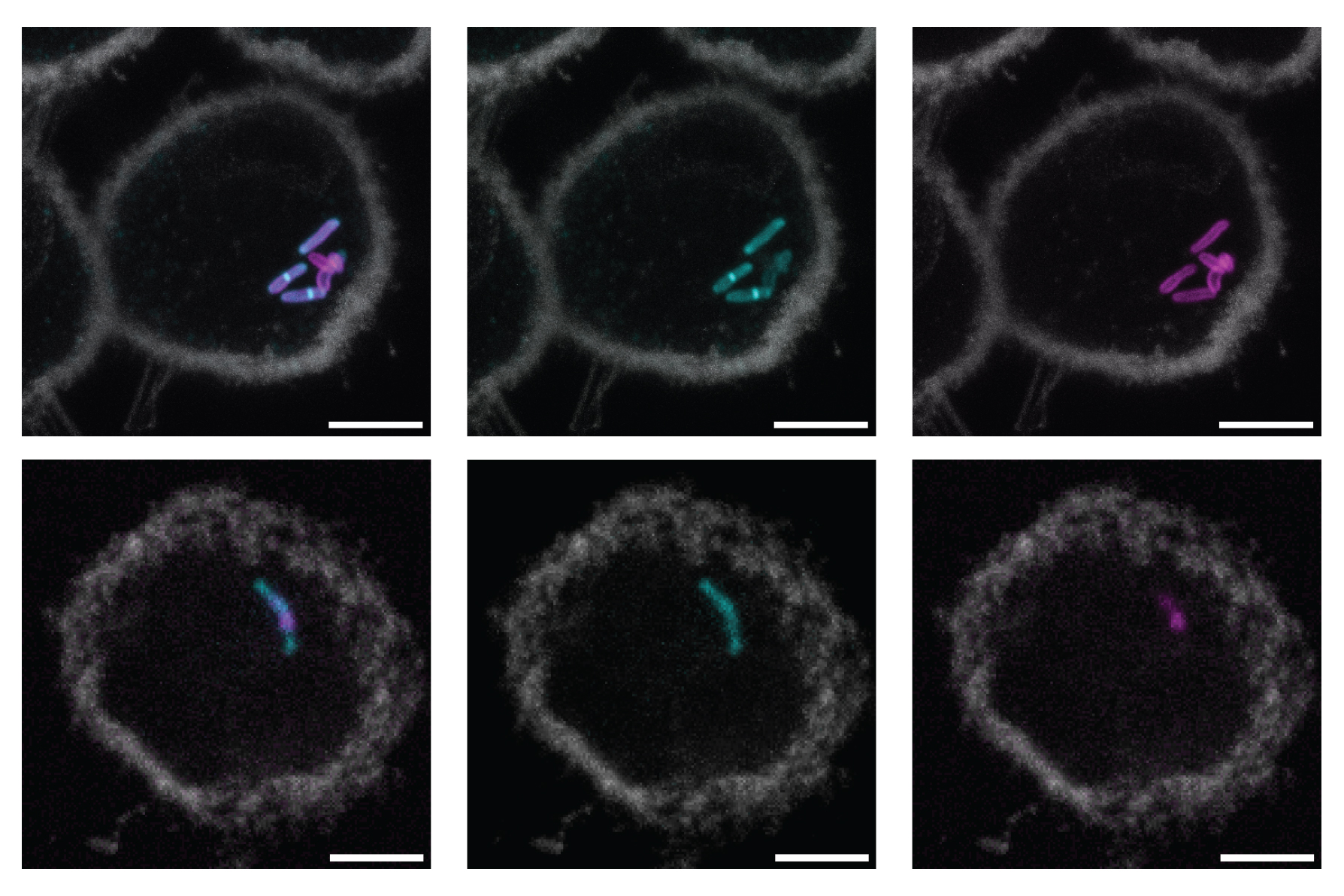
New rifamycin drugs fight antibiotic-resistant lung infections more effectively
Modified antibiotics beat a stubborn lung pathogen and dodge metabolic landmines, offering new hope for patients with HIV, cystic fibrosis, and TB co-infections. Study: Next-generation rifamycins for the treatment of mycobacterial infections. Image Credit …